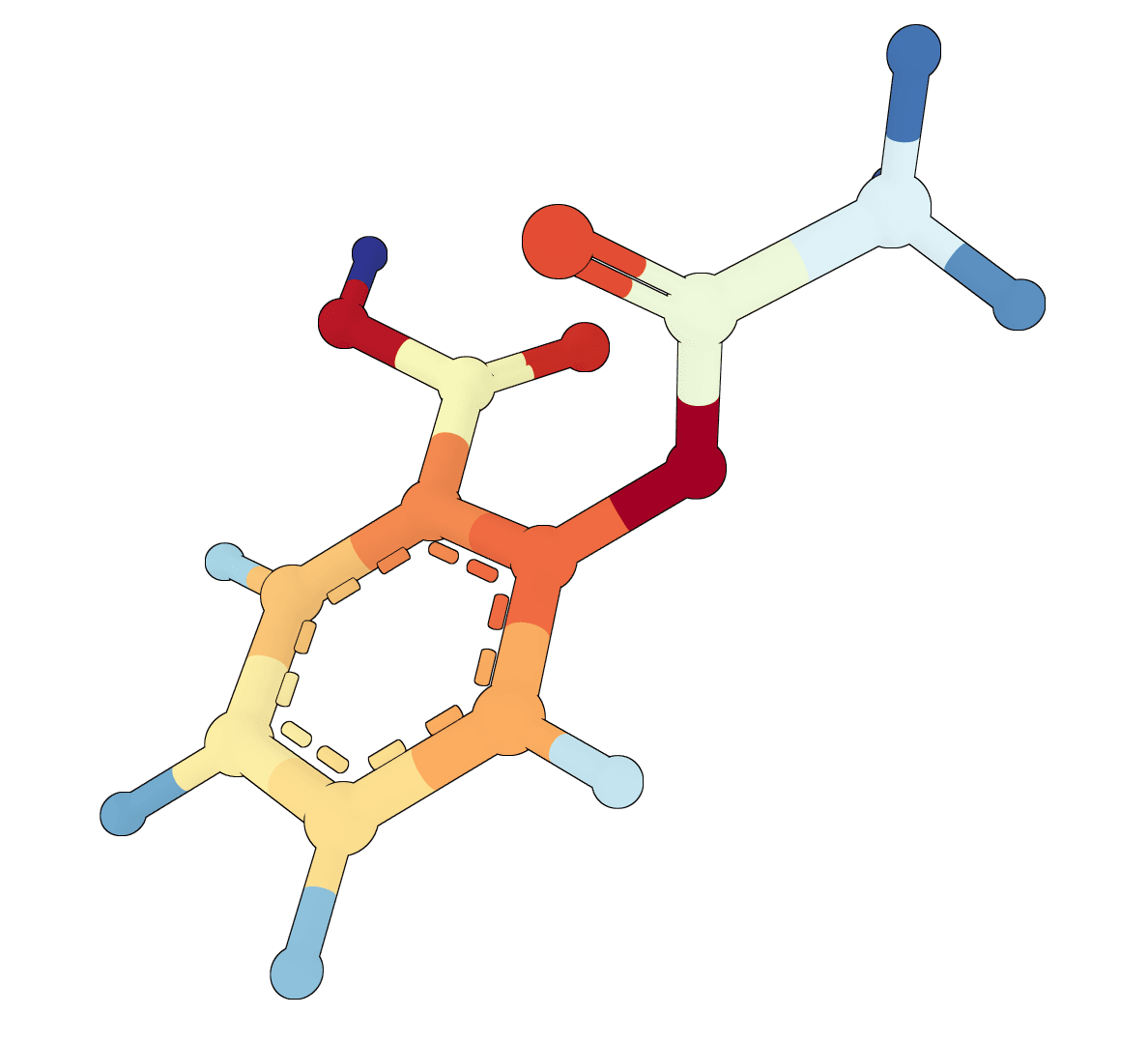
(Single ligand) ADMET-AI aspirin
(Multi mode) ADMET-AI
Related tools
Admetica
Predict 22 ADMET properties from SMILES strings with the upstream Admetica Chemprop models from Datagrok.
eToxPred
Predict toxicity and synthetic accessibility of small molecules using machine learning. eToxPred combines toxicity risk assessment with synthetic accessibility scoring to help prioritize drug candidates.

Brenk filter
Identify toxic, reactive, and pharmacokinetically problematic molecular fragments using structural alert patterns

Lead-likeness filter
Screen for lead-like compounds using stricter molecular descriptor criteria than Lipinski or Veber rules for early-stage drug discovery

PAINS filter
Screen compounds for Pan-Assay INterference patterns that cause false positives in biological assays

QEPPI
Quantitative estimate for protein-protein interaction inhibitor potential. Evaluates drug-likeness for compounds targeting PPIs.
ToxPred 2.0 (Toxicity prediction)
Screen compounds for structural toxicity alerts using PAINS, Brenk, and NIH filters. For focused screening, see PAINS Filter, Brenk Filter, or Veber's Rule.

Veber's rule
Veber's Rule predicts oral bioavailability by evaluating molecular weight, LogP, hydrogen bond donors/acceptors, and rotatable bonds
Lipinski's rule of 5
Lipinski's Rule of Five predicts whether compounds will be orally bioavailable by evaluating molecular weight, LogP, hydrogen bond donors, and acceptors.
SPRINT
SPRINT (Structure-aware PRotein ligand INTeraction) predicts drug-target interactions using co-embedded protein and ligand representations. Screen thousands of compounds against a protein target in seconds.
What is ADMET-AI?
ADMET-AI is a machine-learning system for estimating small-molecule absorption, distribution, metabolism, excretion, and toxicity properties directly from SMILES strings. On ProteinIQ, the current integration runs the ADMET-AI v2 model line with updated training data, updated DrugBank percentile references, and expanded physicochemical alert outputs.
ADMET-AI is typically used during early-stage compound triage, when large libraries need to be prioritized before more expensive synthesis and experimental profiling.
How to use ADMET-AI online
ProteinIQ provides browser-based access to ADMET-AI, so predictions can be generated without local model setup, environment management, or dependency installation.
Inputs
| Input | Description |
|---|---|
SMILES | One or more compounds submitted as plain SMILES (CCO) or name-tab-SMILES (aspirin<TAB>CC(=O)Oc1ccccc1C(=O)O). |
SMILES file | Batch upload via .csv, .tsv, .smi, .smiles, or .txt. |
PubChem fetch | External compound retrieval through the integrated PubChem fetcher. |
Settings
| Setting | Description |
|---|---|
Job name | Optional label for run tracking in job history. |
Outputs
| Output tab | Description |
|---|---|
Results | Spreadsheet with per-compound ADMET predictions, physicochemical properties, structural alerts, and DrugBank percentile columns. |
Files | Generated visual assets, including DrugBank comparison plots, radial ADMET summaries, and molecule structure images. |
Result groups
| Group | Representative columns |
|---|---|
| Compound identity | ids, smiles |
| Physicochemical properties | molecular_weight, logp, hba, hbd, lipinski_violations, qed_score, tpsa |
| Structural alerts | pains_alert, brenk_alert, nih_alert |
| Absorption | hia_absorption, bioavailability, caco2_permeability, pampa_permeability |
| Distribution | bbb_penetration, plasma_protein_binding, volume_distribution |
| Metabolism | CYP inhibitor/substrate endpoints (cyp1a2_inhibitor, cyp3a4_substrate, etc.) |
| Excretion | half_life, clearance_hepatocyte, clearance_microsome |
| Toxicity | herg_blocker, clinical_toxicity, ames_mutagenicity, dili_hepatotoxicity, ld50_toxicity |
| Percentile context | *_percentile columns relative to DrugBank reference distributions |
How does ADMET-AI work?
ADMET-AI v2 uses Chemprop v2 graph neural network models trained across curated ADMET tasks from Therapeutics Data Commons (TDC). Molecules are represented as graphs, and predictions are generated per endpoint across absorption, distribution, metabolism, excretion, and toxicity categories.
Compared with older ADMET-AI v1 deployments, the v2 line replaces the earlier Chemprop v1/Chemprop-RDKit model setup, improves compatibility with modern PyTorch environments, and includes updated training/reference datasets. ProteinIQ also exposes the v2 structural alerts (PAINS_alert, BRENK_alert, NIH_alert) in the default results table.
Interpreting results
ADMET-AI predictions are endpoint-specific and not all columns share the same scale or clinical interpretation. The most reliable workflow is comparative ranking within a project rather than hard-threshold pass/fail filtering from a single endpoint.
Percentile columns
*_percentile columns provide relative context against a DrugBank reference set used by the deployed ADMET-AI resources.
| Percentile range | Typical interpretation |
|---|---|
0-20 | Low relative value vs reference compounds |
20-80 | Mid-range relative value |
80-100 | High relative value vs reference compounds |
Directionality still depends on endpoint semantics. For example, a higher percentile is favorable for some properties and unfavorable for others.
Structural alerts
The v2 alert fields are heuristic flags rather than final exclusion rules.
| Alert column | Interpretation |
|---|---|
pains_alert | Flags substructures associated with assay-interference risk. |
brenk_alert | Flags medicinal-chemistry structural liabilities. |
nih_alert | Flags potentially problematic motifs from NIH filter rules. |
Limitations
- Model scope: ADMET-AI is optimized for small molecules represented in the training domain. Reliability may decrease for chemotypes that are poorly represented in source datasets.
- Endpoint uncertainty: Different ADMET endpoints have different noise levels, assay definitions, and transferability to specific project conditions.
- Comparative use: Predictions support prioritization and hypothesis generation, not standalone go/no-go decisions.
- Version sensitivity: v2 predictions are not numerically identical to v1 predictions because models and reference data were retrained.