Vorobyev S.V., Mizin D.V., Breygina M.A., Bykova E.A., et al.
International Journal of Molecular Sciences· 2026-03· Gubkin University, Moscow, Russia
The research
Auxins are plant hormones that regulate cell division, elongation, and root development. The best-known synthetic auxin, 2,4-dichlorophenoxyacetic acid (2,4-D), has been used as a herbicide and growth regulator for decades. But 2,4-D persists in soil, accumulates in the environment, and lacks selectivity between target weeds and non-target crops.
Researchers at Gubkin University and Lomonosov Moscow State University set out to synthesize a new series of auxin analogs starting from alkylphenols, particularly thymol and related compounds. Alkylphenols introduce a bulkier, more lipophilic aromatic fragment, which prior computational work from the same group had suggested could tune membrane interactions and improve biological activity. The goal was to replace the simple chlorinated phenyl ring of 2,4-D with an alkylated scaffold, then selectively introduce halogen substituents to probe how chlorine, bromine, and iodine each affect activity.
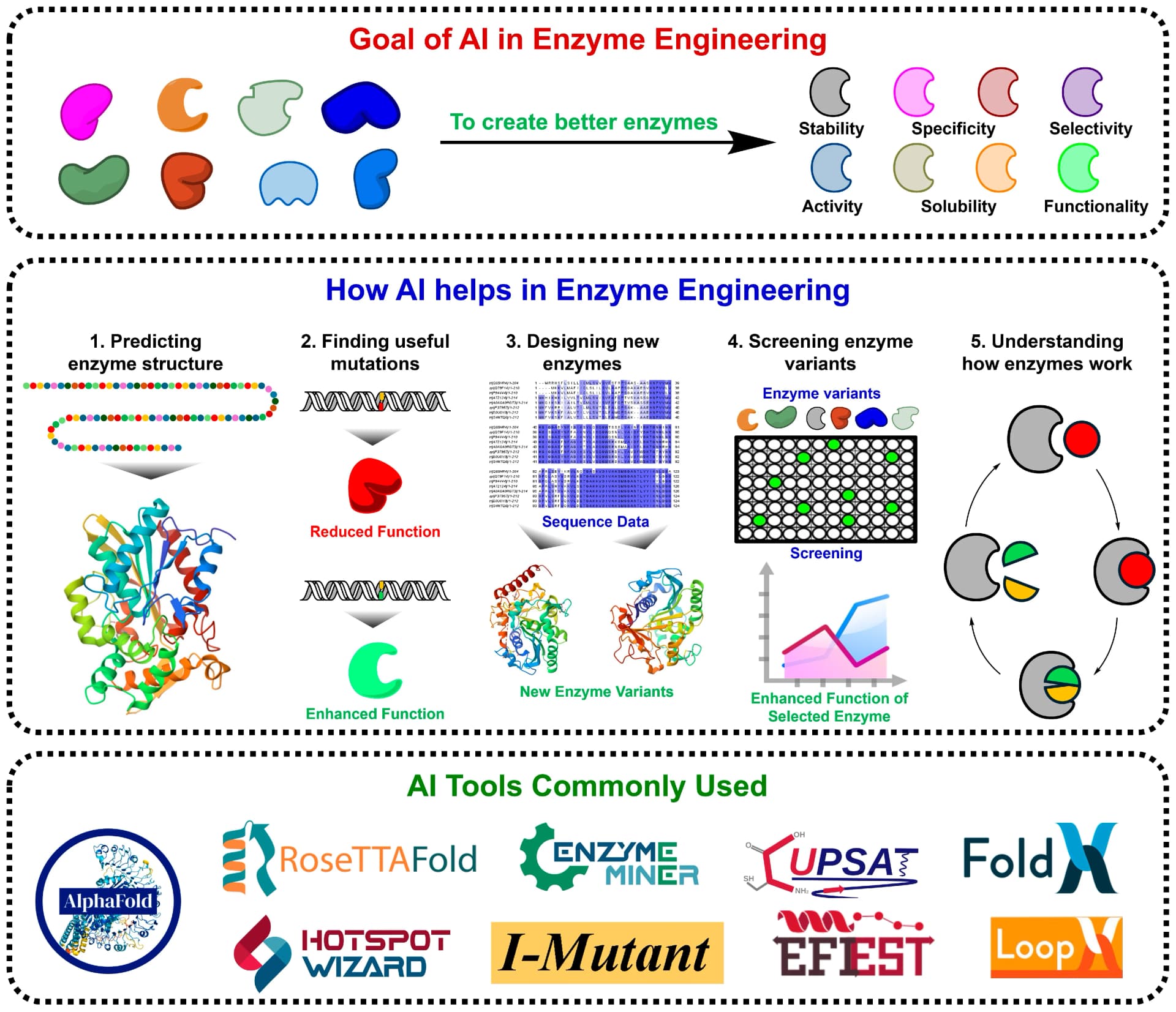
ProteinIQ in action
Synthesizing compounds was half the study. Understanding why certain derivatives were more active required connecting structure to physical properties. Lipophilicity, expressed as logP, is one of the primary parameters researchers use to predict membrane permeability, intracellular transport, and receptor binding. It is also a core input to drug-likeness filters like Lipinski's Rule of 5, making it a natural starting point for characterizing any new compound series.
The team used ProteinIQ's Molecular Descriptors tool to calculate logP for the synthesized alkylphenoxyacetic acids and their halogenated derivatives. The results were compared against the logP of 2,4-D to contextualize how the new structures differ.
One finding from the logP calculations was unexpected: ProteinIQ returned identical logP values for three positional isomers of chloro-dimethylphenoxyacetic acid. The authors noted this directly, describing the result as counter-intuitive. The explanation lies in how the underlying algorithm works. ProteinIQ's logP calculation uses the Wildman-Crippen method, which assigns atomic contributions based on atom type and local chemical environment, then sums them. Because positional isomers share the same atom types and bond patterns, their per-atom contributions are identical, and so is the total. The method does not account for global ring position, only local atomic neighborhoods.
This is a known characteristic of atom-contribution logP methods, not a calculation error. Researchers who need to distinguish positional isomers by lipophilicity can turn to machine-learning-based approaches like ADMET-AI, which learn from experimental training data and can capture subtler structural effects that fragment-based sums miss.
Across the broader compound set, the logP trend was clear: moving from chlorine to bromine to iodine substituents increased lipophilicity, as expected from the heavier halogens. The thymol-derived compounds, which carry an isopropyl group, showed the most pronounced changes. All tested compounds exceeded the logP of 2,4-D, consistent with the alkylphenol scaffold's greater hydrophobicity.
Key findings
In pollen germination assays using tobacco (Nicotiana tabacum L.), all of the halogenated alkylphenoxyacetic acids showed auxin-like stimulating activity. The optimal concentration window was 10^-6 to 10^-7 M, with maximum germination stimulation reaching 157%.
The iodinated thymol derivative, compound 16, produced the strongest response. At 10^-6 M it outperformed all other tested compounds in pollen germination, and its effect was also significant at 10^-7 M, a concentration at which most other analogs showed little or no response. That result contradicts an established expectation in the phenoxyacetic acid literature, where auxin-like activity typically decreases as substituents move from chlorine to bromine to iodine. The authors suggest that prior studies did not use the pollen germination assay, which may be more sensitive to membrane partitioning than classical plant bioassays. Compound 16 also showed lipophilicity an order of magnitude higher than 2,4-D, supporting the hypothesis that membrane incorporation plays a role in its elevated activity.
Seed germination experiments confirmed that compounds 9 and 16 stimulated tobacco seed germination at 10^-6 M, while compound 8 produced the strongest shoot elongation effect in corn seedlings. The differential response between species and assay types points to the complexity of auxin action in plants, and suggests that alkylphenol-derived analogs interact with auxin transport and receptor systems in ways that are not fully captured by existing structure-activity models. For researchers interested in pushing these compounds toward agricultural use, follow-up toxicity screening and ADMET profiling would help assess environmental safety and bioavailability alongside activity. The synthesis route, using N-halosuccinimides or bromine in dioxane, consistently gave yields above 75%, making scale-up straightforward.