MDGen
Generate molecular dynamics trajectories from protein structures.
Related tools
ORB v3
ORB v3 is a universal interatomic potential (machine learning force field) that predicts energies, forces, and stress tensors for atomic systems. Supports both molecular and materials structures with geometry optimization using conservative and direct model variants.
AlphaFlow
Generate protein conformational ensembles with ESMFlow, the single-sequence AlphaFlow model family. Produces multiple diverse structures showing protein flexibility and dynamics.
AlphaFold2
AlphaFold2 via ColabFold for high-accuracy protein structure prediction. Uses MMSeqs2 API for MSA generation with no local databases required. Supports monomer and multimer prediction.
Boltz-2
Boltz-2 is a biomolecular foundation model for structure and binding affinity prediction. Supports proteins, ligands, DNA, and RNA in multi-component complexes. Automatically scales GPU resources for large complexes. Predicts binding affinity with near-FEP accuracy at 1000x faster speed.
Chai-1
Chai-1 is a multi-modal foundation model for molecular structure prediction. Predicts 3D structures for proteins, ligands, DNA, RNA, and multi-component complexes with high accuracy.
ESMfold
ESMfold is a fast, single-sequence protein structure predictor from Meta AI. Predicts 3D protein structures directly from amino acid sequences without requiring multiple sequence alignments (MSA), making it significantly faster than AlphaFold while automatically scaling GPU resources for larger proteins.
GROMACS
Run molecular dynamics simulations using the GROMACS engine with classical force fields (AMBER, CHARMM, GROMOS, OPLS). Study protein dynamics, conformational flexibility, and structural stability with production-grade MD methodology.
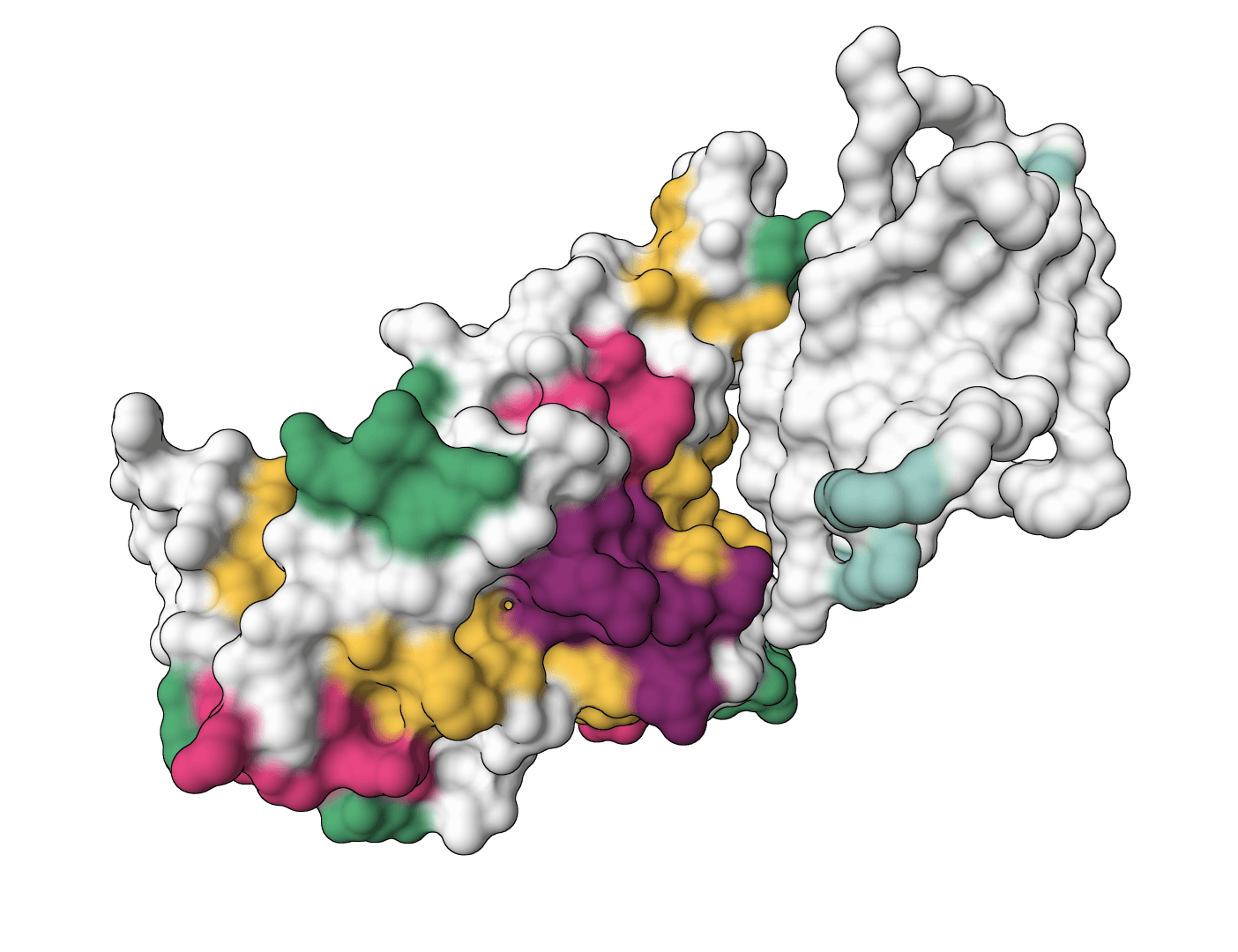
IgGM
IgGM is a generative foundation model for antibody and nanobody design against a target antigen. Supports CDR design, affinity maturation, inverse design, and framework design. Requires an antigen structure (PDB) and antibody sequences with "X" marking positions to design.
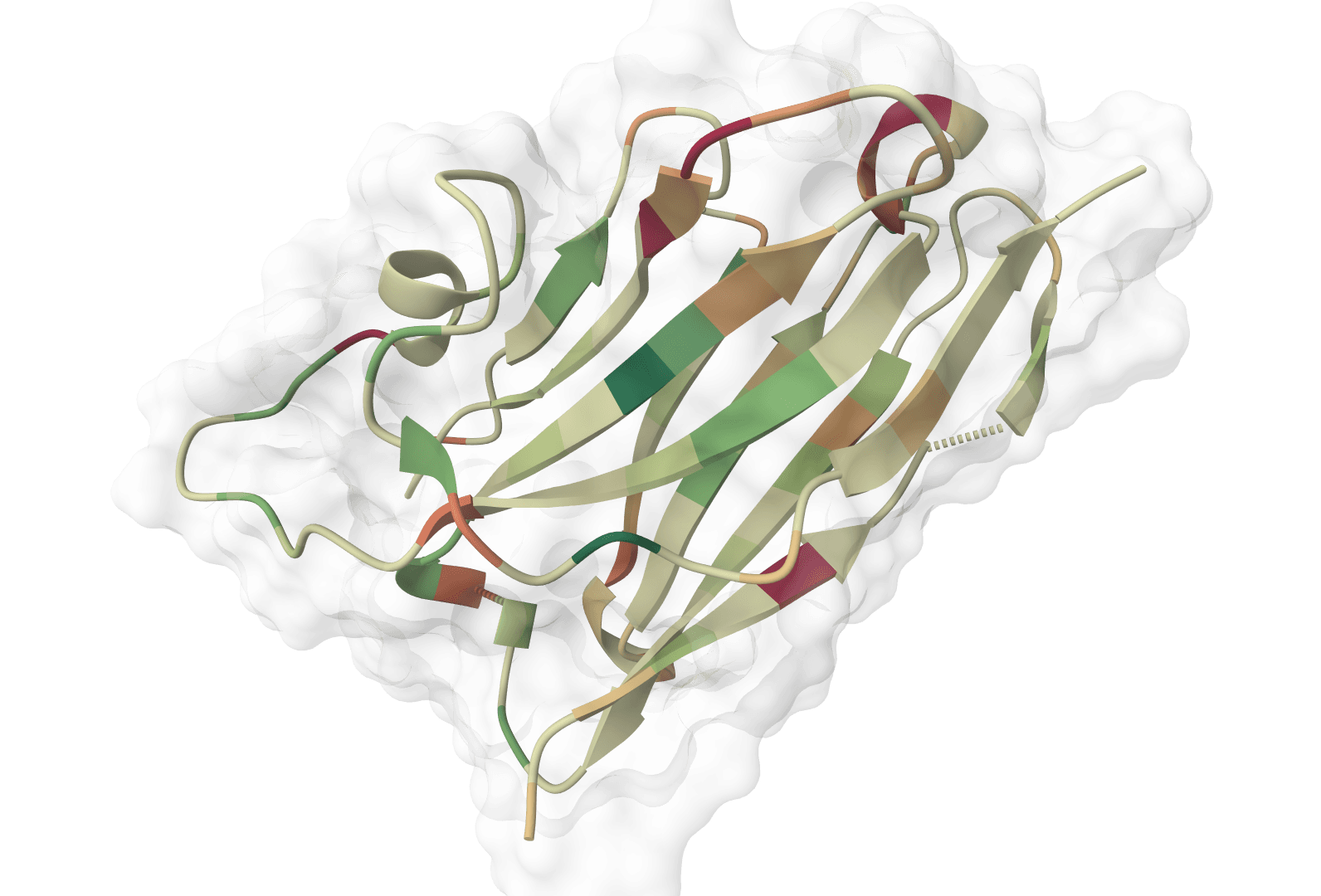
ImmuneBuilder
ImmuneBuilder predicts 3D structures of immune receptor proteins including antibodies, nanobodies, and T-cell receptors. It uses ABodyBuilder2, NanoBodyBuilder2, and TCRBuilder2/TCRBuilder2+ to generate structures with per-residue error estimates and optional ensemble artifacts.
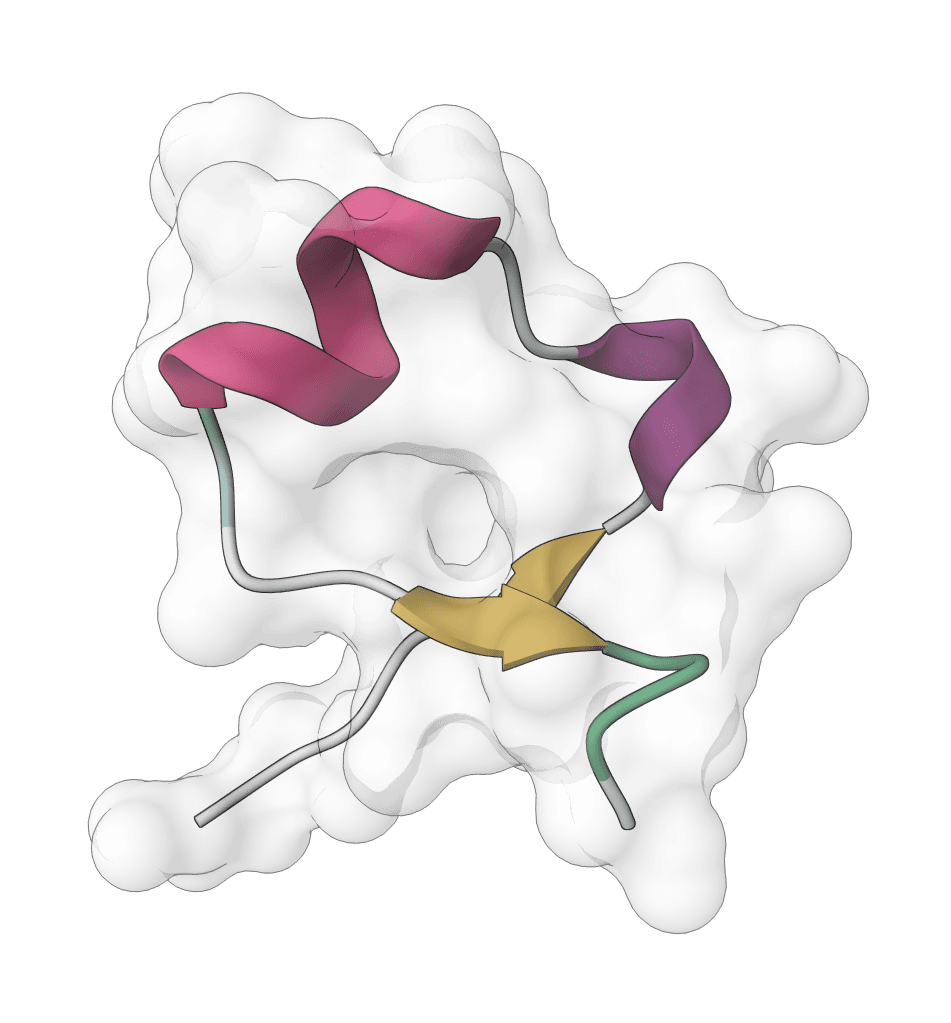
IntelliFold 2
Controllable biomolecular structure prediction model for proteins, ligands, DNA, RNA, and multi-component complexes. IntelliFold 2 supports fast v2-Flash inference, optional MSA generation, and ranked confidence outputs.
What is MDGen?
MDGen generates molecular dynamics trajectories using generative AI rather than physics-based simulation. Given a single protein structure, it produces a sequence of conformations representing how the protein might move over time—achieving speedups of 10–1000× compared to traditional MD while preserving key dynamic properties.
The model learns from molecular dynamics simulation data to capture realistic protein motions. Unlike physics-based simulators that integrate equations of motion at femtosecond timesteps, MDGen directly generates trajectory frames, making it practical to explore conformational ensembles in seconds rather than days.
How MDGen works
MDGen frames trajectory generation as a conditional generative modeling problem. The model is trained on molecular dynamics simulation data and learns to generate plausible time evolutions by conditioning on trajectory frames.
Architecture
The system uses a Scalable Interpolant Transformer (SiT) as its flow-based generative backbone. This avoids the computationally expensive residue-pair and frame-based architectures common in protein structure prediction. To handle long trajectories, MDGen incorporates the Hyena long-context architecture, enabling scaling to trajectories of 100,000+ frames.
Proteins are represented in the atom14 format (14 atoms per residue) and converted to SE(3) rigid frames (translation + rotation) plus torsion angles. This representation captures both backbone geometry and sidechain conformations.
Training data
MDGen provides checkpoints trained on different datasets:
- Tetrapeptides: Explicit and implicit solvent simulations of 4-residue peptides, used for method validation
- ATLAS: The ATLAS dataset of protein monomer simulations, preprocessed to 400 picosecond intervals, enabling generation for full proteins
Supported tasks
The generative approach enables multiple tasks through different conditioning strategies:
| Task | Description |
|---|---|
| Forward simulation | Generate trajectory from an initial structure |
| Transition path sampling | Given start and end states, sample plausible connecting paths |
| Trajectory upsampling | Increase temporal resolution of existing trajectories |
| Inpainting | Generate partial molecular dynamics conditioned on fixed regions |
How to use MDGen online
ProteinIQ hosts MDGen on GPU infrastructure with pre-loaded model weights, generating trajectories directly in the browser.
Input
| Input | Description |
|---|---|
Protein Structure | PDB file, mmCIF file, or PDB ID (e.g., 1AKI). Maximum 1,000 residues. |
Settings
Trajectory parameters
| Setting | Description |
|---|---|
Number of frames | Trajectory length (10–100, default 50). More frames capture longer timescale dynamics but increase computation. |
Sampling temperature | Diversity control (0.1–2.0, default 1.0). Lower = more conservative motions, higher = more exploration. |
Advanced options
| Setting | Description |
|---|---|
Frame stride | Save every Nth frame (1–10, default 1). Higher values reduce output size. |
Random seed | Fixed seed for reproducibility. Leave empty for random sampling each run. |
Output
MDGen produces a trajectory viewable in the integrated 3D viewer:
| Output | Description |
|---|---|
| Topology PDB | Reference structure with atom connectivity information |
| Trajectory XTC | Compressed trajectory file containing all frames |
| RMSD metrics | Average and maximum backbone deviation from the starting structure |
The viewer supports playback controls, frame-by-frame navigation, and structure alignment.
When to use MDGen vs traditional MD
MDGen excels at rapid conformational exploration when physical accuracy is less critical than speed:
| Use case | MDGen | Traditional MD |
|---|---|---|
| Quick conformational screening | Fast sampling across multiple proteins | Computationally prohibitive |
| Qualitative dynamics exploration | Reasonable ensemble diversity | Higher accuracy needed |
| Large-scale studies | Practical for hundreds of proteins | Resource-intensive |
| Binding site flexibility | Rapid estimate of accessible conformations | Detailed energetics needed |
For applications requiring accurate free energy estimates, specific timescale information, or force field validation, physics-based MD remains the appropriate choice.
Limitations
MDGen is designed for research exploration and has several constraints:
- Protein size: Best results for proteins under 256 residues (ATLAS training limit); larger proteins may produce less reliable dynamics
- Physical accuracy: Generated trajectories approximate, but do not exactly reproduce, true molecular dynamics
- Timescales: The model captures conformational diversity but not absolute timescale information
- Ligands and cofactors: Currently supports protein-only trajectories; bound ligands are not handled
Interpreting results
RMSD values
Root-mean-square deviation measures how much the structure changes from the starting conformation:
| RMSD (nm) | Interpretation |
|---|---|
| < 0.1 | Minimal backbone motion, local fluctuations only |
| 0.1–0.3 | Moderate conformational change, typical for stable proteins |
| 0.3–0.5 | Significant rearrangement, loop movements or domain shifts |
| > 0.5 | Large-scale conformational change |
Trajectory quality
Evaluate generated trajectories by checking:
- Continuity: Frames should show smooth transitions without sudden jumps
- Physical plausibility: No steric clashes, bond lengths should remain reasonable
- Diversity: Multiple conformational states should be sampled